Journal: ACS Sens. 2019, 451329-1336
Authors: George Papadakis, Alexandros K. Pantazis, Maria Ntogka, Konstantinos Parasyris, Gesthimani-Ioanna Theodosi, Georgia Kaprou, Electra Gizeli
Abstract: The objective of this work is to develop a methodology and associated platform for nucleic acid detection at the point-of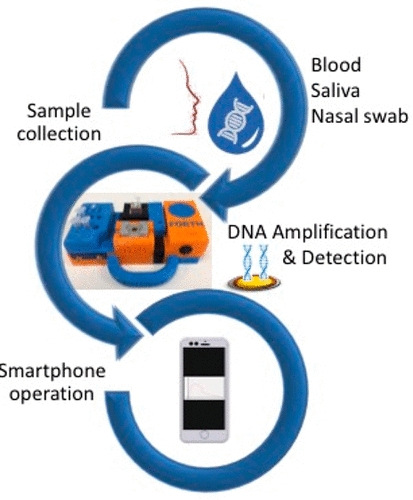 -care (POC) that is sensitive, user-friendly, affordable, rapid, and robust. The heart of this system is an acoustic wave sensor, based on a Surface Acoustic Wave (SAW) or Quartz Crystal Microbalance (QCM) device, which is employed for the label-free detection of isothermally amplified target DNA. Nucleic acids amplification and detection is demonstrated inside three crude human samples, i.e., whole blood, saliva, and nasal swab, spiked in with 10–100 Salmonella cells. To qualify for POC applications, a portable platform was developed based on 3D printing, integrating inside a single box: (i) simple fluidics based on plastic tubing and a mini peristaltic pump, (ii) a heating plate combined with disposable reaction tubes for isothermal amplification; (iii) a mini antenna analyzer operated through a tablet; and (iv) an acoustic wave device housing unit. The simplicity of the method combined with smartphone operation and detection, rapid sample-to-answer analysis time (30 min), and high performance (detection limit 4 × 103 CFU/ml) in three of the most important human samples in diagnostics suggest that the methodology could become a tool of choice for nucleic acid detection at the POC. In addition, the low cost of the platform and assay holds promise for its adoption in resource limited areas. The acoustic detection method is shown to give similar results with a standard colorimetric assay carried out in saliva and nasal swab but can also be used to detect nucleic acids inside whole blood, where a colorimetric assay failed to perform.
-care (POC) that is sensitive, user-friendly, affordable, rapid, and robust. The heart of this system is an acoustic wave sensor, based on a Surface Acoustic Wave (SAW) or Quartz Crystal Microbalance (QCM) device, which is employed for the label-free detection of isothermally amplified target DNA. Nucleic acids amplification and detection is demonstrated inside three crude human samples, i.e., whole blood, saliva, and nasal swab, spiked in with 10–100 Salmonella cells. To qualify for POC applications, a portable platform was developed based on 3D printing, integrating inside a single box: (i) simple fluidics based on plastic tubing and a mini peristaltic pump, (ii) a heating plate combined with disposable reaction tubes for isothermal amplification; (iii) a mini antenna analyzer operated through a tablet; and (iv) an acoustic wave device housing unit. The simplicity of the method combined with smartphone operation and detection, rapid sample-to-answer analysis time (30 min), and high performance (detection limit 4 × 103 CFU/ml) in three of the most important human samples in diagnostics suggest that the methodology could become a tool of choice for nucleic acid detection at the POC. In addition, the low cost of the platform and assay holds promise for its adoption in resource limited areas. The acoustic detection method is shown to give similar results with a standard colorimetric assay carried out in saliva and nasal swab but can also be used to detect nucleic acids inside whole blood, where a colorimetric assay failed to perform.
Keywords: acoustic biosensor, LAMP, quantitative nucleic acid detection, crude human samples, point-of-care Salmonella testing, smartphone, molecular diagnostics, resource limited areas.


