Synthesis and covalent immobilization of redox-active metallopolymers for organic phase electrochemistry
Authors: Hanna Hübner, Riccardo Candeago, Deborah Schmitt, Alexander Schießer, Beichen Xiong, Markus Gallei and Xiao Su.
Journal: Polymer (2022) 244, 124656.
Abstract
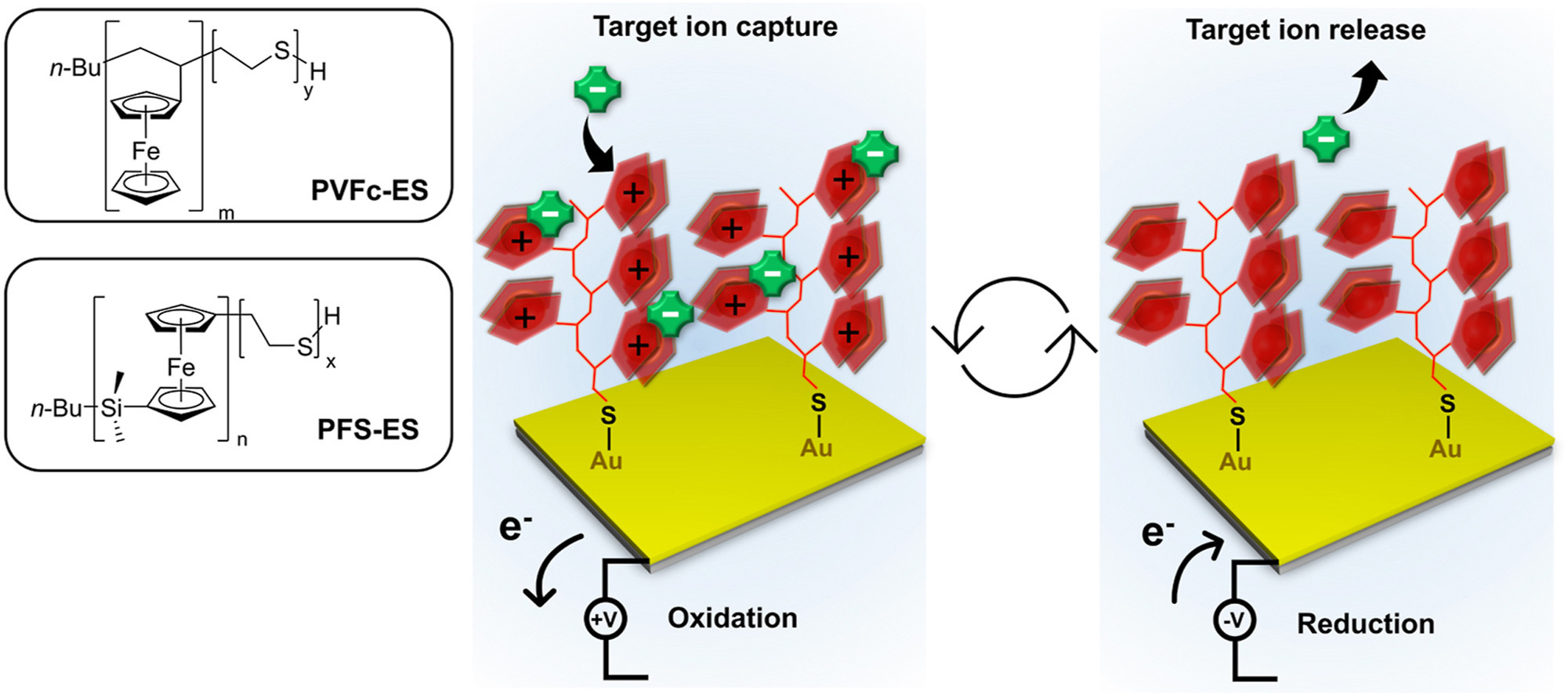
Redox-active metallopolymers are promising stimuli-responsive platforms for a range of applications including sensing, energy storage, and selective separations. In particular, heterogeneously-functionalized metallopolymers can modulate the capture and release of target molecules, driven by redox electron-transfer. However, prior metallopolymer-functionalized electrodes have been fabricated by non-covalent methods, and tailored for aqueous phase applications. As such, despite the existing potential for heterogeneous applications in organic phase, there are significant constraints to the stability of metallopolymers in organic solvents, including high solubility in solvents such as chloroform or tetrahydrofuran. We propose the immobilization of thiol-functionalized redox-active metallopolymers on metallic surfaces as a facile way to enhance stability and cyclability in organic media, and thus broaden the applicability of redox-metallopolymers for organic phase applications. We explore the anionic polymerization of metal-containing monomers vinylferrocene (VFc) and ferrocenyldimethylsilane (FS), and their thiol end-functionalization by living anionic polymerization strategies. PFS and PVFc with molar masses ranging from 1800 to 49900 g mol−1 and 2900 to 6300 g mol−1 respectively were prepared with a segment of poly(ethylene sulfide), as characterized by size-exclusion chromatography, NMR spectroscopy, MALDI/ToF, thermogravimetry, and elemental analysis. Both metallopolymers were immobilized on gold substrates by a grafting-to protocol, with demonstrated redox-responsiveness by electrochemical control. In the case of immobilized PVFc, operando electrochemical testing demonstrated the stable and reversible electrochemical cycling capabilities (>74% maximum current retained after 100 oxidation/reduction cycles) in several organic solvents including chloroform, tetrahydrofuran, ethanol, methanol, acetonitrile, and acetone. Immobilized PFS was stable in chloroform, with a 83% maximum current retained after 100 oxidation/reduction cycles. We envision future applications of these covalently immobilized metallopolymers for a broad range of fields from selective separations to sensing and energy storage.
You may read the full paper here.
Synthesis and covalent immobilization of redox-active metallopolymers for organic phase electrochemistry



